Researchers at Agriculture Victoria compared three non-destructive instruments against a destructive penetrometer to test the accuracy of the instruments, and determine whether non-destructive measurements were a suitable replacement for penetrometer measurements of flesh firmness.
Measuring changes in stone fruit ripeness during fruit growth is an important step to help determine when growers need to harvest and guide them with their postharvest handling and marketing strategy. A key quality trait that is universally accepted as a relatively accurate measure of ripeness is flesh firmness (FF) and is traditionally measured using a handheld penetrometer or pressure tester, with measurement units of kg per cm2. This instrument is portable and easy to use in the field, cool room or packhouse. However, measurements on the same fruit can vary depending on the operator, the measurements are destructive, and high fruit numbers are generally required to estimate the average firmness within a batch of fruit. In recent years several instruments have been developed and commercialised which can non-destructively measure changes in firmness to help identify the optimal time for harvest. Utilising these novel instruments requires lower fruit numbers, with tested fruit still saleable after measurement.
Research scientists: Glenn Hale and John Lopresti
Advantages:
- portable
- ease of use
- non-destructive measurements
Standard penetrometer flesh firmness measurements were conducted using an electronic desk-top fruit texture analyser (FTA) where a constant penetration speed and depth were used to minimise operator error (Figure 1.1). Penetrometer flesh firmness measurements from this instrument were compared to two non-destructive hand-held firmness meters, an Agrosta®100 USB (Figure 1.2), and a Turoni hardness tester (Figure 1.3). A DA meter that non-destructively measures changes in chlorophyll concentration directly under the skin surface via an index of absorbance was also included in the comparison (Figure 1.4).
Figure 1 photos: Instruments used to measure quality changes in stone fruit.
 Fruit Texture Analyser
Fruit Texture Analyser
Figure 1.1 Fruit texture analyser (photo: gusstoday).
 Agrosta firmness meter
Agrosta firmness meter
Figure 1.2 Agrosta firmness meter (photo: agro-technologies)
 Turoni meter
Turoni meter
Figure 1.3 Turoni hardness tester (photo: turoni)
 DA meter
DA meter
Figure 1.4 DA meter (photo: turoni)
Ripeness measurements were conducted during a postharvest storage experiment using three stone fruit cultivars. ‘Majestic Pearl’ white nectarines were harvested from a commercial orchard in Swan Hill and ‘September Bright’ yellow nectarines harvested from Agriculture Victoria’s SmartFarm in Tatura, whilst ‘Sunset’ plums were sourced directly from an exporter in Melbourne, Victoria (Figure 2).
Figure 2. Initial fruit quality of ‘Majestic Pearl’ nectarine, ‘Sunset’ plum and ‘September Bright’ nectarine prior to cool storage.
Fruit was initially assessed at harvest and then stored for four weeks at 2 °C followed by a ripening period of up to seven days at 18 °C, with two or three assessments conducted during ripening. The linear relationship between each instrument was calculated using the coefficient of determination (r2).
The strength of the linear relationship between measurements of firmness varied for each nectarine and plum. For example, in ‘Majestic Pearl’ nectarine, a good correlation (r2 = 0.60) was observed between the FTA and the Agrosta instrument (Table 1). In other words, Agrosta measurements explained 60 % of the variation in flesh firmness as measured by the FTA.
Table 1. Coefficient of determination (r2) between three non-destructive firmness devices (DA meter, Agrosta firmness meter and Turoni hardness tester), and standard penetrometer flesh firmness measurements using the destructive fruit texture analyser instrument for ‘Majestic Pearl’ nectarine at harvest, after cool storage and during ripening.
FTA | DA meter | Agrosta | |
|---|---|---|---|
DA meter | 0.31 | ||
Agrosta | 0.60 | 0.34 | |
Turoni | 0.42 | 0.39 | 0.49 |
The DA meter measurements of chlorophyll concentration were not well correlated with firmness measurements (i.e., r2 = 0.01 to 0.39), and should not be used to estimate flesh firmness in these three cultivars. The DA meter has previously been shown to have a higher correlation with fruit ethylene production and hence fruit physiological maturity, but not flesh firmness. Among non-destructive instruments and the three cultivars, the best correlation was observed between the Agrosta firmness meter and the FTA unit (Figure 3).
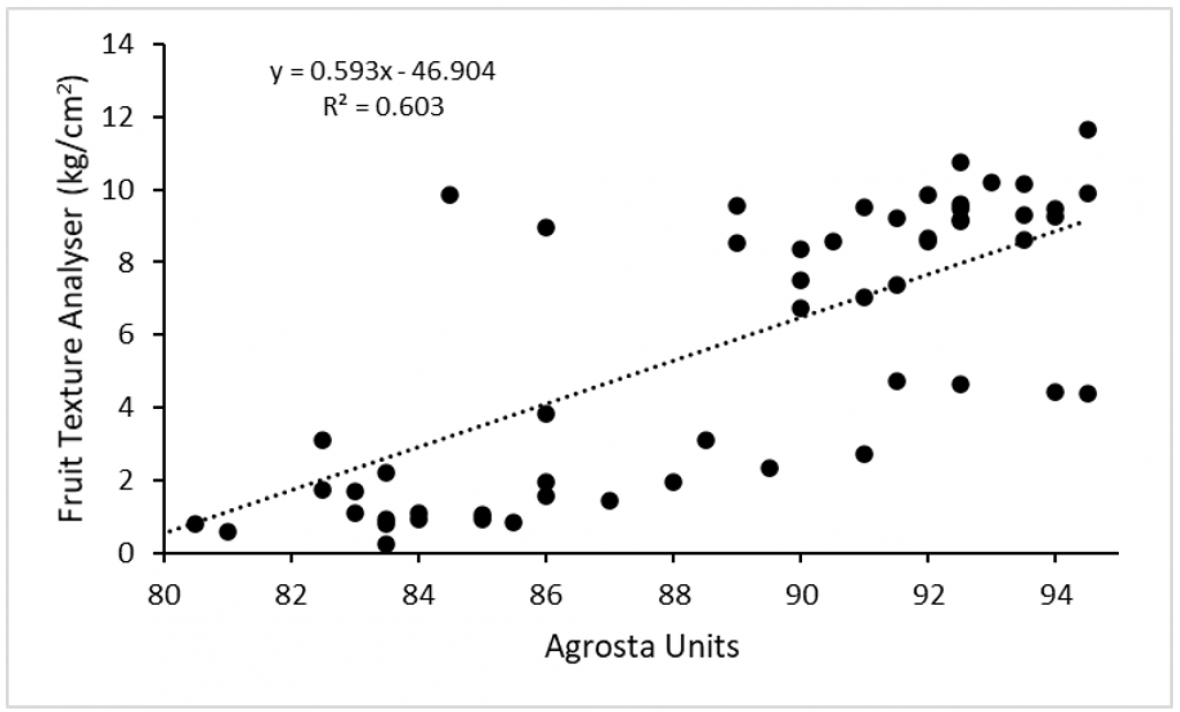
Figure 3. Relationship of standard penetrometer flesh firmness measurements using the destructive fruit texture analyser with an Agrosta firmness meter for ‘Majestic Pearl’ nectarine at harvest, after cool storage and during ripening.
The handheld Agrosta meter successfully measured the change in ‘Majestic Pearl’ firmness between approximately 82 units (mature fruit) to 93 units (immature fruit) which equates to a destructive measurement with a penetrometer of 2 kg/cm2 and 8 kg/cm2, respectively (Figure 3).
The Agrosta device has several advantages over current penetrometer instruments such as its portability enabling use in the orchard on fruit prior to harvest, during cool storage or in the packhouse. Another advantage is that the same fruit on the tree or after harvest can be monitored for changes in ripening due to its ability to non-destructively measure flesh firmness, allowing a more accurate understanding of fruit ripening behaviour pre- and post-harvest.
Acknowledgements
The Serviced Supply Chains project is funded by the Hort Frontiers Asian markets Fund, part of the Hort Frontiers Asian strategic partnership initiative developed by Hort Innovation, with co-investment from Agriculture Victoria, the Department of Agriculture and Fisheries Queensland (DAFQ), Montague Fresh (summerfruit), Manbulloo (mangoes), Glen Grove (citrus), the Australian Government plus in-kind support from University of Queensland and the Chinese Academy of Sciences.


